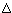 fH°
fH°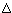 fG°
fG°| Beryllium Bromide (BeBr2) | Be1Br2(cr) | |||||||||||||
| Enthalpy Reference Temperature = Tr = 298.15 K | Standard State Pressure = p° = 0.1 MPa | |||||||||||||
| J·K-1 mol-1 | kJ·mol-1 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T/K | Cp° | S° | -[G°-H°(Tr)]/T | H-H°(Tr) | 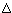 fH° fH° |
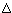 fG° fG° |
log Kf | |||||||
| 0 | ||||||||||||||
| 100 | ||||||||||||||
| 200 | ||||||||||||||
| 298.15 | 66.024 | 100.416 | 100.416 | 0. | -355.640 | -337.384 | 59.108 | |||||||
| 300 | 66.149 | 100.825 | 100.417 | 0.122 | -355.688 | -337.271 | 58.724 | |||||||
| 400 | 70.626 | 120.475 | 103.066 | 6.963 | -385.169 | -324.995 | 42.440 | |||||||
| 500 | 74.810 | 136.710 | 108.216 | 14.247 | -383.680 | -310.120 | 32.398 | |||||||
| 600 | 77.613 | 150.608 | 114.151 | 21.874 | -382.039 | -295.560 | 25.731 | |||||||
| 700 | 79.705 | 162.735 | 120.244 | 29.744 | -380.299 | -281.283 | 20.990 | |||||||
| 800 | 81.337 | 173.487 | 126.240 | 37.798 | -378.495 | -267.260 | 17.450 | |||||||
| 900 | 82.634 | 183.144 | 132.035 | 45.998 | -376.651 | -253.467 | 14.711 | |||||||
| 1000 | 83.764 | 191.909 | 137.591 | 54.318 | -374.788 | -239.879 | 12.530 | |||||||
| 1100 | 84.810 | 199.942 | 142.899 | 62.747 | -372.913 | -226.479 | 10.755 | |||||||
| 1200 | 85.856 | 207.366 | 147.966 | 71.280 | -371.030 | -213.250 | 9.283 | |||||||
| 1300 | 86.902 | 214.280 | 152.804 | 79.919 | -369.137 | -200.178 | 8.043 | |||||||
| 1400 | 87.864 | 220.755 | 157.429 | 88.656 | -367.241 | -187.252 | 6.986 | |||||||
| 1500 | 88.910 | 226.852 | 161.856 | 97.494 | -365.343 | -174.462 | 6.075 | |||||||
| PREVIOUS: June 1965 | CURRENT: June 1975 | |||||||||||||
| Beryllium Bromide (BeBr2) | Be1Br2(cr) | |||||||||||||
Download table in tab-delimited text format.