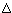 fH°
fH°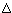 fG°
fG°| Sodium (Na) | Na1(l) | |||||||||||||
| Enthalpy Reference Temperature = Tr = 298.15 K | Standard State Pressure = p° = 0.1 MPa | |||||||||||||
| J·K-1 mol-1 | kJ·mol-1 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T/K | Cp° | S° | -[G°-H°(Tr)]/T | H-H°(Tr) | 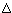 fH° fH° |
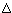 fG° fG° |
log Kf | |||||||
| 0 | ||||||||||||||
| 100 | ||||||||||||||
| 200 | ||||||||||||||
| 250 | ||||||||||||||
| 298.15 | 32.706 | 57.862 | 57.862 | 0. | 2.406 | 0.495 | -0.087 | |||||||
| 300 | 32.685 | 58.064 | 57.863 | 0.060 | 2.414 | 0.483 | -0.084 | |||||||
| 350 | 32.068 | 63.056 | 58.258 | 1.679 | 2.580 | 0.147 | -0.022 | |||||||
| 370.980 | 31.827 | 64.915 | 58.583 | 2.349 | CRYSTAL <--> LIQUID | |||||||||
| 400 | 31.510 | 67.301 | 59.130 | 3.268 | 0. 0. 0. | |||||||||
| 450 | 31.004 | 70.982 | 60.247 | 4.831 | 0. | 0. | 0. | |||||||
| 500 | 30.552 | 74.225 | 61.486 | 6.370 | 0. | 0. | 0. | |||||||
| 600 | 29.807 | 79.726 | 64.083 | 9.386 | 0. | 0. | 0. | |||||||
| 700 | 29.271 | 84.278 | 66.652 | 12.338 | 0. | 0. | 0. | |||||||
| 800 | 28.945 | 88.163 | 69.104 | 15.247 | 0. | 0. | 0. | |||||||
| 900 | 28.836 | 91.564 | 71.415 | 18.134 | 0. | 0. | 0. | |||||||
| 1000 | 28.945 | 94.606 | 73.584 | 21.021 | 0. | 0. | 0. | |||||||
| 1100 | 29.259 | 97.377 | 75.623 | 23.930 | 0. | 0. | 0. | |||||||
| 1170.525 | 29.613 | 99.206 | 76.989 | 26.005 | FUGACITY = 1 bar | |||||||||
| 1200 | 29.790 | 99.945 | 77.544 | 26.881 | 96.760 2.440 0.106 | |||||||||
| 1300 | 30.514 | 102.357 | 79.361 | 29.895 | -95.824 | 10.669 | -0.429 | |||||||
| 1400 | 31.351 | 104.649 | 81.086 | 32.988 | -94.809 | 18.823 | -0.702 | |||||||
| 1500 | 32.188 | 106.840 | 82.730 | 36.165 | -93.711 | 26.902 | -0.937 | |||||||
| 1600 | 33.024 | 108.944 | 84.303 | 39.426 | -92.529 | 34.905 | -1.140 | |||||||
| PREVIOUS: | CURRENT: June 1962 | |||||||||||||
| Sodium (Na) | Na1(l) | |||||||||||||
Download table in tab-delimited text format.